Mepitel in paediatric burns
Prospective, randomised study of the efficacy of Mepitel® on children with partial-thickness scalds
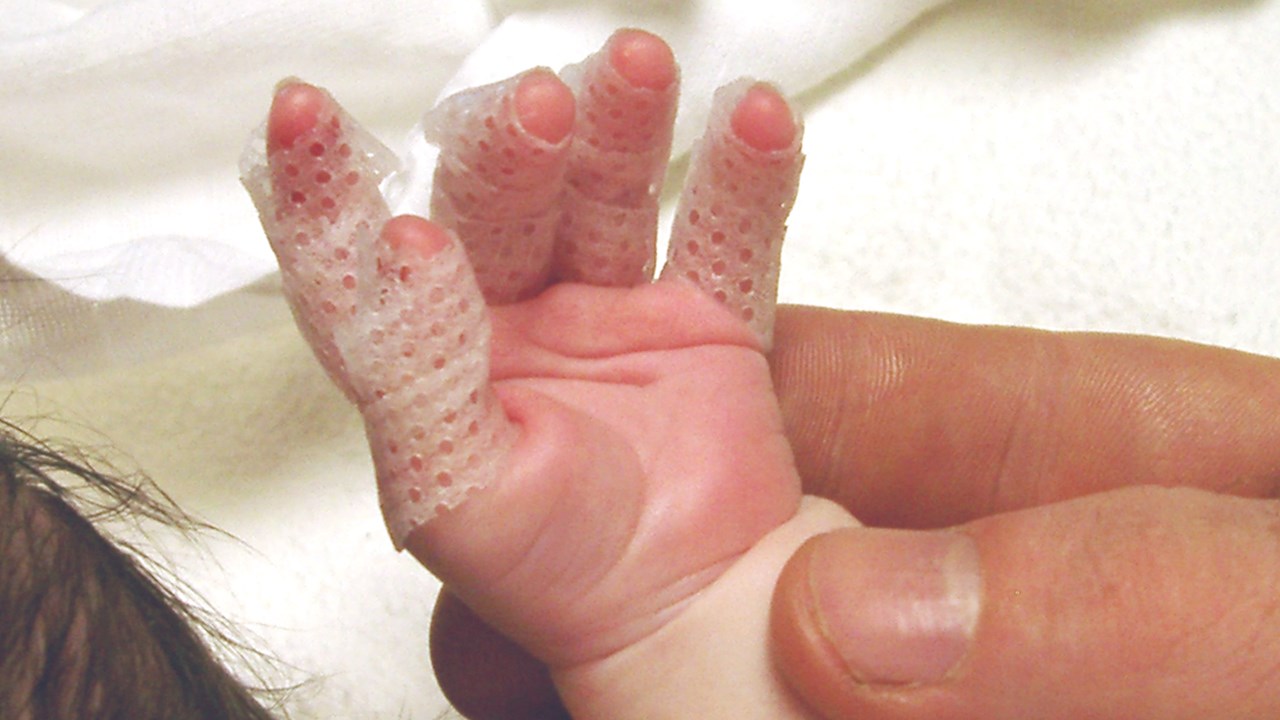
Aim:
This paper presents the results of a randomised controlled clinical trial that was undertaken to evaluate the efficacy of Mepitel in 63 children with partial-thickness scald burns.
Methods:
Children aged 12 years and younger admitted with partial-thickness scald burns covering 15% total body surface area (TBSA) were randomly assigned to treatment with either Mepitel or silver sulfadiazine (SSD). Secondary wet and dry under-cotton gauze dressings were applied over both treatments.
Data were collected on time to wound healing (as measured by the number of days until the wounds were 25%, 50%, 75% and 100% epithelialised), eschar formation, pain at dressing change (measured using objective pain scale) and the time required for the dressing change.
After the patient was discharged, billed hospital charges were analysed to determine dressing-related charges, narcotics charges and total charges.
Result:
Wounds of children treated with Mepitel healed significantly faster (p>0.001), exhibited less eschar formation (p>0.05) and experienced less pain at dressing change (p>0.05), than those in the control group.
The children treated with Mepitel had approximately 75% fewer dressing changes than those in the control groups, thus further reducing the pain experienced by the children and resulting in significantly lower charges for dressing changes and narcotics.
Patients treated with Mepitel were also associated with significantly lower mean daily hospital charges (US$1937 vs US$2316: p=0.025).
Conclusions:
Mepitel exhibited superior healing times and reduced pain levels during dressing changes as compared to treatment with SSD. Children treated with Mepitel underwent approximately 75% fewer dressing changes then did those treated with SSD. The authors believe the use of Mepitel represents a significant advance in the treatment of partial-thickness scald burns in children.
Expertise
Knowledge'References'






